What does an Fda approval suggest for cosmetic treatments and units?
Evaluate Prices of Beauty Medical procedures in France with British isles Costs
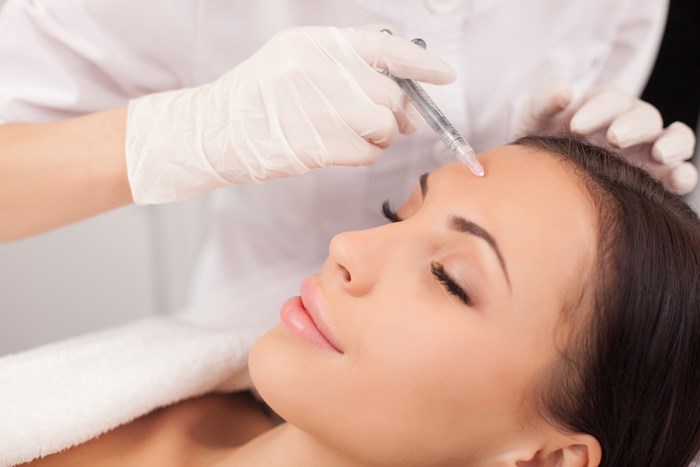
Late 2017 brought a great deal of interest to BOTOX®. In St. Louis, the place the group of plastic surgeons at Washington College uses the injectable to smooth out crows’ ft and far more, the information of a new approval from the Foodstuff and Drug Administration (Food and drug administration) was celebrated as it was close to the rest of the place. The acceptance was for a cosmetic use – minimizing the look of horizontal brow traces linked with action in the frontalis muscle mass.
Although this sort of approvals are schedule for the countrywide administration, they can also elevate questions amongst the common community. What does an Food and drug administration acceptance suggest? How is it attained? And what if a healthcare service provider or other health-related specialist suggests some thing that isn’t really approved by the Food and drug administration?
The U.S. Food and drug administration regulates foods (aside from aspects of eggs and meat dealt with by the U.S. Division of Agriculture), vaccines, medicines for people and animals, tobacco goods, nutritional health supplements, radiation-emitting electronics, cosmetics and medical products for human beings. People final two include the bulk of what is accessible at a standard plastic surgical treatment office and incorporate lasers, liposuction and other body fat reduction products, injectables (not just BOTOX®, but other botulinum toxin-based treatments, as well as dermal fillers and Kybella®for double chins), breast implants (regarded to be units) and a lot more.
Virtually fifty percent (forty eight%) of the approvals manufactured by the Fda are for health-related devices. A single 3rd (33%) are presented to foods intended for human consumption. The following premier slice of the pie is cosmetics (eight%), followed by housewares and meals-relevant objects at five p.c, and drugs and “biologics” at 3%.
The administration’s aim is to protect the individuals and animals of the United States from harm by ensuring that the relevant meals and medication are marketed in approaches constant with what they have been verified to properly attain. For case in point, a yogurt maker cannot claim that its food is a miracle cure for male pattern baldness (except if, of training course, adequate trials and studies demonstrate that it is).
Fundamentally, these regulations avert merchandise and gadget-makers from creating wild or otherwise unsubstantiated promises to increase revenue or in any other case motivate individuals to purchase what they are offering in the hopes of obtaining a specific outcome.
This also means that healthcare pros are totally free to use these goods in other techniques. Even though BOTOX®is U.S. Fda-accepted only for a few cosmetic uses—frown traces, crow’s toes and now horizontal brow lines—the injectable is routinely employed at West County Surgeons of Washington University and elsewhere to do every little thing from clean out bunny strains on the nose to improve the visual appeal of the lips to allow downward-dragged eyebrows to sit in a more normally calm and uplifted place.
These utilizes, although not formally acknowledged by the Food and drug administration, even so have a historical past of productive and safe use, constructed by suppliers close to the region. Occasionally, this kind of common applications—called “off-label utilizes”—can direct to an Food and drug administration approval soon after many years of medical trials and cautiously executed reports. The body of knowledge created by healthcare pros as a collective entire can reward patients in the interim, as effectively as provide as a launching level for new traces of study and inquiry.
Although Food and drug administration approvals happen in a assortment of methods, depending on what’s up for approval, the route that drugs and health-related units get is a demanding a single. Businesses need to prove their choices to be equally secure and effective before they can be put on the market place. This includes medical screening, usually overseen by medical pros acquainted with the item or device in issue or comparable goods and units.